Aytu BioScience, Inc. (NASDAQ:AYTU), a specialty pharmaceutical company focused on commercializing novel products that address significant patient needs announced today that the Company has signed an agreement to distribute the Pinnacle CovID RAD Rapid Antigen Detection Test worldwide. The rapid antigen test, which delivers results in fifteen minutes, tests for the presence of the SARS-CoV-2 virus antigen via a nasopharyngeal sample and can be conducted without the use of laboratory equipment.
Pinnacle IVD Corporation plans to scale U.S. manufacturing capacity for the CovID RAD Rapid Antigen Detection Test to 25 million tests per month. Source Aytu BioScience
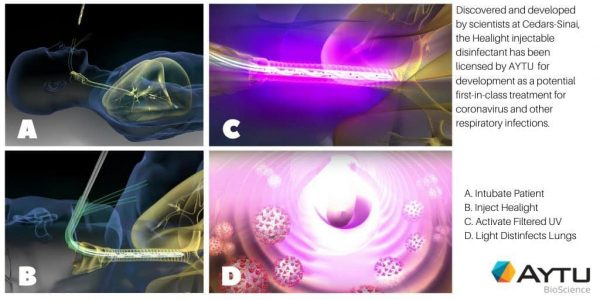
Aytu BioScience, Inc. (NASDAQ:AYTU) (the “Company”), a specialty pharmaceutical company focused on commercializing novel products that address significant patient needs announced today that it has signed an exclusive worldwide license from Cedars-Sinai to develop and commercialize the Healight Platform Technology (“Healight”). This medical device technology platform, discovered and developed by scientists at Cedars-Sinai, is being studied as a potential first-in-class treatment for coronavirus and other respiratory infections. Source: AYTU
Ultraviolet (UV) Light
Ultraviolet (UV) is a type of electromagnetic radiation with wavelengths from 10 nm to 400 nm. These wavelengths are shorter than that of visible light. Between the wavelengths 100 to 400 nm ultraviolet radiation (UV light) is subcategorized into three different ranges: Ultraviolet C (UVC) 100 — 280 nm, Ultraviolet B (UVB) 280 — 315 nm, and Ultraviolet A (UVA) 315 — 400 nm.
Spectrum of Light
UVC light is weak at the Earth’s surface since it is absorbed by the ozone layer of the atmosphere, however UVC from manufactured lamps/lights has been widely used as a commercial germicide. Radiation between the 200 nm and 300 nm wavelengths are strongly absorbed by nucleic acid (DNA & RNA), leading to nucleic acid damage, and resulting in inactivation of the organism or death.
While UVC light has broad germicidal properties, it is also harmful to mammalian (human) cells. Alternatively, UVA and UVB devices have been FDA-approved with indications to treat human diseases including skin lymphoma, eczema, and psoriasis. Of the three spectrums, UVA light appears to cause the least damage to mammalian cells. Recent advances in light emitting diodes (LEDs) have made it much more feasible to manufacture and apply narrow band (NB) UVA light to internal organs.
Proof of Concept
An abstract led by the team at Cedars-Sinai Medical Center was published in the United European Gastroenterology Journal, October 2019, titled “Internally Applied Ultraviolet Light as a Novel Approach for Effective and Safe Anti-Microbial Treatment.” Here, the authors show that UVA light exhibits significant in vitro bactericidal effects in an array of clinically important bacteria. Additionally, this is the first study using intracolonic UVA application, which reports that UVA exposure is not associated with endoscopic or histologic injury. These findings suggest that UVA therapy can potentially provide a safe and effective novel approach to antimicrobial treatment via phototherapy on internal organs.
https://www.ueg.eu/education/document/internally-applied-ultraviolet-light-as-a-novel-approach-for-effective-and-safe-anti-microbial-treatment/208958/
Analyst Price Target on AYTU $3.00 â–² (152.10% Upside) Based on 1 analyst offering 12-month price targets for Aytu BioScience in the last 3 months. The average price target is $3.00 with a high forecast of $3.00 and a low forecast of $3.00. The average price target represents a 152.10% increase from the last price of $1.19.Source: TipRanks
Wall Street is positive on Aytu Bioscience Inc (AYTU). On average, analysts give Aytu Bioscience Inc a Strong Buy rating. The average price target is $2.75, which means analysts expect the stock to gain by 169.61% over the next twelve months.Source Investors Corner
Positive trends in the stock include: Revenue 3 Year (CAGR) 63.36% and Gross Profit Margin 74.95%
On Coronavirus impact, we see fewer negative headwinds to the drug and managed-care industries as patients prioritize drug therapy and avoid costly elective surgeries that can weigh on managed-care margins. Conversely, we expect increased negative pressure to the device, dental, life sciences, and hospital industries as patients avoid in-office visits and hospitals reduce capacity to perform high-margin elective procedures. Source Charles Schwab Research Morning Star Report PDF AYTU
The company has these products in their pipeline:
Therapeutics
ACIPHEX ® SPRINKLEâ„¢ (rabeprazole sodium) is a granule formulation of rabeprazole sodium, a commonly prescribed proton pump inhibitor. AcipHex Sprinkle is indicated for the treatment of gastroesophageal reflux disease (GERD) in pediatric patients 1 to 11 years of age for up to 12 weeks.
CEFACLOR (cefaclor oral suspension) is a second-generation cephalosporin antibiotic suspension and is indicated for the treatment of numerous common infections caused by Streptococcus pneumoniae, Haemophilus influenzae, staphylococci, and Streptococcus pyogenes, and others.
FLEXICHAMBER ® is an anti-static, valved collapsible holding chamber intended to be used by patients to administer aerosolized medication from most pressurized metered dose inhalers (MDIs) such as commonly used asthma medications.
KARBINAL ® ER (carbinoxamine maleate extended-release oral suspension) is an H 1 receptor antagonist (antihistamine) indicated to treat various allergic conditions including seasonal and perennial allergic rhinitis, vasomotor rhinitis, and other common allergic conditions.
NATESTO ® (testosterone) nasal gel, the only FDA-approved nasally administered testosterone indicated for replacement therapy in adult males with a deficiency or absence of endogenous testosterone due to primary hypogonadism.
POLY-VI-FLOR ® and TRI-VI-FLOR ® are two complementary prescription fluoride-based supplement product lines containing combinations of vitamins and fluoride in various oral formulations. These prescription supplements are prescribed for infants and children to treat or prevent fluoride deficiency due to poor diet or low levels of fluoride in drinking water and other sources.
TUZISTRA ® XR (Codeine Polistirex and Chlorpheniramine Polistirex) is the only codeine-based extended-release oral suspension cough-cold treatment in the US prescription cough-cold market.
ZOLPIMISTâ„¢ (zolpidem tartrate) Oral Spray, the only FDA-approved oral spray zolpidem product indicated for the short-term treatment of insomnia characterized by difficulties with sleep initiation.
Diagnostics
THE MiOXSYS ® SYSTEM A novel, rapid in vitro diagnostic semen analysis system with the potential to become a standard of care in the diagnosis and management of male infertility.
Aytu’s strategy is to continue building its portfolio of revenue-generating Rx and consumer health products, leveraging its focused commercial team and expertise to build leading brands within large therapeutic markets. For more information visit aytubio.com and visit innovuspharma.com to learn about the company’s consumer healthcare products.
CWEB Analysts see the stock as a potential for long and short term growth with huge rewards in the post and current pandemic conditions and upwards of $5 by 2021
